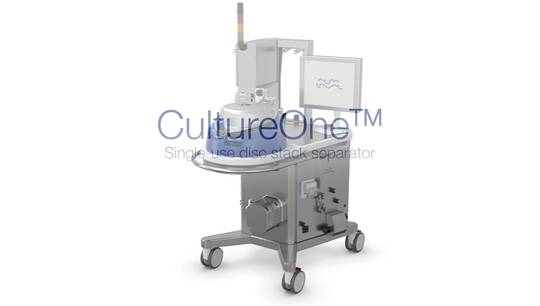
HSS
Premium separator for single-use biopharmaceutical processing
1,117 views
With the new Alfa Laval CultureOne, all of our unique innovations for gentle treatment and increased yield are now available in a solution for single-use biopharma processing.
CultureOne brings maximum separation efficiency while creating new possibilities for processing cell-culture-based fermentations.
View transcript
Welcome. My name is Anette Holfve and I’m your host today. In April last year, we launched Alfa Laval CultureOne to the single-use biopharma industry. And now, more than a year later, after multiple trials at multiple customer sites we want to share the findings and how we are delivering value to our customers. With me in studio to share these findings, I have three of my dear colleagues: Doug Osman, who is our Director for Life Sciences here at Alfa Laval. Doug has worked many years at Alfa Laval and has been part of this development from the start. Mikaela Sifvert, who is our Global Launch Manager. She will share our white paper findings that we did together with a customer. Third, but not least, Peter Thorwid, who is our Concept Development Manager in our R&D department and has of course been part of this development from the start. Doug, we start with you! In case we have people joining this webinar who have not listened to the previous ones would you mind giving a short recap of what we launched in April last year and what our starting point was? In April of 2020, we launched the CultureOne: the first premium single-use separator for the harvesting of biotherapeutics. We saw a need in this category, as an extension of our existing stainless steel product line in order to deal with the evolved materials that our customers have been producing. A lot of upstream advances have resulted in higher drug concentrations higher cell concentrations, and the existing solutions for harvesting this material were really being pushed to the limit. We responded to a market demand for something that would deal with these changed and evolved materials. We also looked further into the future to make sure that the solution we generated would be applicable for even the next generation of changes that we see our customers are pursuing. That, in summary, was the motivation behind producing the CultureOne – both from being able to fit into a single-use environment, but also to deal with the changes to the materials from the upstream processes. In developing an approved solution against those process challenges what are the most important criteria? First and foremost for our customers is product yield. Any recovery method must deliver a high product yield in this first step, because losses in this first step have a multiplicative effect in terms of the overall yield from the efforts that our customers make to produce these materials. Then, we’re looking at how the quality of the materials that we produce from this first step affects the downstream operations: various filter and purification operations downstream. We wanted something that delivered a high-quality material after the separation result. Finally, we were looking at something that would treat the material quite gently so that we would not introduce additional impurities that would have to be dealt with downstream as well. These three criteria – yield, downstream filterability and low additional lyses – were the primary considerations in terms of delivering a good result out of this particular process step. Any additional consideration that we brought into the development? We wanted something that would operate on a continuous basis, so that we could reach a steady state and have something run in a predictable and reliable way. We also wanted something that would deliver this process result without any additives. Additives complicate operations, and they add expense and time and risk. We wanted something that would operate without the need for any chemical addition. We also wanted something that would be quite simple to operate as well. A lot of these operations involve portable equipment. Having something that would operate quite easily and simply in that type of environment is very important to making sure that the results that are achieved are repeatable every time. Those were some of the additional considerations that we looked at in terms of having something that would function well in this type of environment. After a year working closely with the customers, what is the feedback that you hear, Doug? Some of the things that we’re hearing back are exactly what we hoped to hear. We do have something that delivers a fantastic process result, that is simple and intuitive to work with, that produces repeatable and reliable results, and really delivers on the things that customers value in this particular process step. We’ve heard other comments such as: “It treats the material quite gently.” “It’s a very quiet system, it doesn’t generate a lot of noise and is easy to work right next to.” “It’s portable and straightforward to work with, and really kind of intuitive, and provides fantastic feedback in terms of what is going on while you’re making a process run.” We’ve had a lot of good comments back, and it truly has been great to hear that, because it has validated that we have delivered against the original design criteria. Thank you, Doug! What I hear you say is that we took departure in the problems that customers are facing when we created the solution, and the feedback we get is very, very positive. That’s good to hear. Thank you! We also have documented results of the performance that we can deliver. We published a white paper late last year together with a customer. Mikaela, welcome! Would you mind telling us a little about this initiative? During the development of CultureOne, we had a very strong partner in Genentech one of the leading biopharmaceutical companies, based in California. Together, we ran numbers of tests. A lot of trials. We did that on both commercial cell culture material and their products under development under real-world conditions. During the tests, we looked at key process factors like yield, filterability additional lyses, and the result turned out to be very good. Together with Genentech, we published these results. We wrote a white paper that was recently published by Bioprocess International. You wanted to highlight three things from the white paper. You talk about product yield, low additional lyses, filterability -- which is also what Doug mentioned earlier and said is important to the customer. If we take it one-by-one, and start with product yield what does this mean and what results did we see? The product yield is the percentage of valuable proteins, so this is a real key factor. For all the tests that we did, under all conditions, we were above our target of 90% product yield. We have also run further tests and reached up to 98%. So, that result is very good. The secret behind it – the reason for this – is basically that the centrifugal technology is a very efficient separation technology. With the right rotation speed plus the high separation area with the latest disc stack design we get the reason behind this result. Secondly, we wanted to highlight the low additional lyses. Could you tell us a little bit about what that means, and also the results that we saw? Additional lyses is the amount of cells that get damaged through a process step. In our case, with CultureOne, we saw that lysed cells were typically very low well below the target. Even at high flow rates, we saw that the additional lyses levels were as low as 5%. This result is typically connected to the optimal rotation speed and the minimized shear on the fluid, which you get with the TopStream design. Finally, we wanted to highlight filterability. Can you tell us what this means and also what results we have documented? Filterability, or filtration throughput, is the filtering characteristic of the fluid after centrifugation. This is essential for the downstream processing, what happens after the separation step. And that includes sterile filtration. Our tests showed that we can achieve as high as 900 l/m2 in filterability. And those results were actually comparable or better than what we see for any baseline process. The key, again, is very efficient centrifugal separation, or removal of cells and debris, which is connected to the Hermetic Design and the TopStream. What you mentioned now is the results published in the white paper that we did together with Genentech. Would you say that these results are stand-alone, or have we been able to repeat them? We’ve had continuous proof of concept for CultureOne. We have continued to document these levels of performance, and we have repeated them with more than a dozen of our customers by now. In my mind, the R&D team, together with Genentech, during this concept development has taken us a great step further. CultureOne is a unique, fine-tuned design concept based on a lot of test results, proving that Hermetic Design, TopStream and UniDisc can really contribute to a consistent and efficient cell harvesting process. Thank you, Mikaela! The white paper shows we have very high product yield. I hear you mention as high as 98%, which is really good, and that the customer product has very high quality after the separation. For me as a non-biopharma-expert, it sounds like the customers get a high-quality product and more of it in this separation process. That’s good! Thank you! Welcome Peter! You are our Concept Development Manager at our R&D department, and you have been part of this development from the start. Can you share a little bit about the approach that we have had to product development in this process? It’s been an agile approach together with the customer. We have, already from the first meeting and the first field test, modified our test centrifuge – the Explorer – and made it into a single-use device. We went to the customer, and we tried some of the new features, blending with some of the knowledge we have from the past. Since the cells that we are separating are not readily available, there is no test media we had to understand how it actually worked in real life. Early prototyping and step-by-step work with the customer toward a final product was the approach and something we succeeded with. Why could we do this? It’s because we didn’t do everything new. It looks different, but it’s still the same separation principles that Alfa Laval has used for decades. Hermetic Technology, together with better disc performance with the UniDisc and also our new development called TopStream, where we treat the cell-heavy phase in a different way. That’s the development we started a few years back, and now when this possibility came up and this need from the customer, we blended the technologies together and could approach the development task in quite a speedy way and to the point. It’s blending new with old – in a new package. What would you say is the next step? It’s to continue on the path that we have set out, continuing to work with the customers, understanding what’s around the corner, what their future needs are. At the same time, showing them what is possible to do. Now that we have a collaboration, we know the prototyping works in their industry we can then show new values and other ways of using our technology to solve their future problems. In the collaboration with the customer a lot of exciting things can happen when we work together. And we’re really looking forward to the future. We’re here to stay. Thank you, Peter! In summary, we have taken departure in separation technology that we know well, we have blended unique Alfa Laval technologies that we know deliver results and we have created something completely new. This is great! Doug, I have one final question for you. Can the customer now use this product in the GMP production environment? Yes. One of the most important criteria for the customers is to have process-contact surfaces which can be validated against the appropriate regulatory authorities in different markets around the world. We have done all this work: all the laboratory studies, all the sterility studies, against it. We’re happy to say that the CultureOne is very well suited for operation in the GMP production environment. Thank you to Doug, Mikaela and Peter for joining us today. And thank you for listening in to this webinar. If you would like to know more about our product and what we can do for you, please reach out to your local Alfa Laval office in the country where you are. Or you can check this page out, fill in the form, and we will guide you to the right person. Thank you again – and stay safe and healthy!